Phosphatidylcholine restores neuronal plasticity of neural stem cells under inflammatory stress
Dario Magaquian, Susana Delgado Ocaña, Consuelo Perez & Claudia Banchio�
Instituto de Biología Molecular y Celular de Rosario (IBR, CONICET) Ocampo y Esmeralda, Predio CONICET and Departamento de Ciencias Biológicas, Facultad de Ciencias Bioquímicas y Farmacéuticas, Universidad Nacional de Rosario, 2000 Rosario, Argentina.
www.nature.com/scientificreports
The balances between neural stem cell (NSCs) growth and differentiation, and between glial and neuronal differentiation play a key role in brain regeneration after any pathological conditions. It is well known that the nervous tissue shows a poor recovery after injury due to the factors present in the wounded microenvironment, particularly inflammatory factors, that prevent neuronal differentiation. Thus, it is essential to generate a favourable condition for NSCs and conduct them to differentiate towards functional neurons. Here, we show that neuroinflammation has no effect on NSCs proliferation but induces an aberrant neuronal differentiation that gives rise to dystrophic, non-functional neurons. This is perhaps the initial step of brain failure associated to many neurological disorders. Interestingly, we demonstrate that phosphatidylcholine (PtdCho)-enriched media enhances neuronal differentiation even under inflammatory stress by modifying the commitment of post-mitotic cells. The pro-neurogenic effect of PtdCho increases the population of healthy normal neurons. In addition, we provide evidences that this phospholipid ameliorates the damage of neurons and, in consequence, modulates neuronal plasticity. These results contribute to our understanding of NSCs behaviour under inflammatory conditions, opening up new venues to improve neurogenic capacity in the brain.
Despite its diverse presentation, inflammation is a common feature across several neuropathological processes and has been implicated as a critical mechanism responsible for the progression of neurodegenerative disorders including Parkinson’s disease, Alzheimer’s disease, multiple sclerosis1–3 as well as traumatic brain injury4,5 and stroke2,6,7. Neuroinflammation is considered a double-edged sword, with protective as well as detrimental effects on the nervous system, especially during repair and recovery. In response to different types of injuries that cause neurons and oligodendrocytes death, activated astrocytes and the resident immune-like glial cells, the micro-glia, proliferate and generate proinflammatory cytokines (such as IL-1, IL-6, IFN-γ and TNF-α), chemokines, prostaglandins, and free oxygen radicals, often leading to the development of cerebral damage, and promoting macrophages infiltration8. Both kinds of cells act as a host defence mechanism eliminating cellular debris and releasing inflammatory factors. These factors finally activate astrocytes, which proliferate and form the glia scar to define a dense limiting border between the healthy and damaged tissue. Two major niches of neural stem cells (NSCs) that support neurogenesis are in the subventricular zone and in the dentate gyrus of the hippocampus of the adult mammalian brain9,10. NSCs are multipotent self-renewing cells that have a regenerative potential because they can proliferate, migrate and differentiate into neurons, astrocytes or oligodendrocytes and thus, promote functional and structural repair of the injured tissue. Several studies have evidenced a cross-talk between immune modulators and NSCs fate11–13. In response to inflammatory reactions, it was shown that the glia scar could prevent tissue regeneration by NSCs8, and that LPS-induced neuroinflammation caused synapse loss by a mechanism dependent of microglia activation and IL-1β secretion14. In this scenario, understanding the NSCs response to these conditions and the mechanisms involved in the integration into the injured brain will be critical for the development of effective therapeutic strategies using stem cells.
We have previously demonstrated that phospholipids affect the fate of post-mitotic neural precursors; specifi-cally, phosphatidylcholine (PtdCho) promotes neuronal differentiation at expenses of astroglial and unspecified precursors15. As the loss of neurons is the detrimental outcome of brain injuries and neurodegenerative diseases, we asked whether PtdCho could still favour neuronal differentiation under inflammatory conditions, and thus prevent or restore tissue damage in this context. By different approaches we have demonstrated that under pro-inflammatory culturing conditions there is an increase in neuronal differentiation that could support a renewal mechanism needed for tissue reparation. Strikingly, under the same conditions, neurons also display an aberrant morphology that could reflect the deleterious effect of neuroinflammation. Interestingly, addition of liposome of egg-source PtdCho further induces neuronal differentiation and also rescues the morphological and functional deficit by modulating neuronal plasticity.
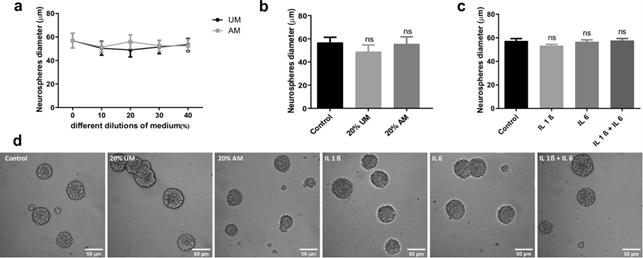
Figure 1. NSCs proliferation is not affected by inflammation. (a) After incubating the NSCs with different dilutions of medium obtained from LPS-stimulated macrophages (AM) or without stimulation as a control (UM) during 96 h, proliferation was analysed by measuring Neurosphere’s diameter. Graph represents the neurosphere’s diameter measured in three independent experiments. (b) Diameter of the Neurospheres of NSCs exposed to 20% V/V of UM and AM or control during 4 days. Graph represents the neurosphere’s diameter measured in three independent experiments. ns no statistical significance. (c) Diameter of the Neurospheres of NSCs exposed to 50 ng/ml of IL-1β and/or IL-6 during 96 h. Graph represents the neurosphere’s diameter measured in three independent experiments. ns no statistical significance. (d) Representative images (× 20) of neurospheres incubated in the indicated conditions. Scale bars: 50 μm.
Results
Effect of inflammatory stress on NSCs proliferation. The balance between NSCs proliferation and differentiation is essential for tissue repair16 and up to know is not clear how it is affected by inflammation. To that end, we incubated NSCs under normal proliferative condition (in the presence of EGF and FGF, neuro-sphere culture) supplemented with different concentrations (% V/V) of macrophages-activated media (AM) or with media without activation as a control (UM). We confirmed by RT-PCR that macrophages activated with LPS express and, as a consequence, secrete IL-1β, IL-6 and TNF-α to the media as previously demonstrated17,18 (Supplementary Fig. 1A). Treated cells were incubated for 4 days, and after this time, cell viability was analysed by MTT assay; UM plus LPS was also included to evaluate LPS toxicity as control (Supplementary Fig. 1B,C). To induce a moderate stress, cells were treated with AM (20% V/V) and, after 4 days, the neurosphere’s diameter was measured as a growth parameter (Fig. 1b). As Fig. 1a,b,d show, there is no significant change in the prolifera-tion rate in the presence of different concentrations (V/V) of activated media respect to media without activation and control. To evaluate the role of each IL individually, similar analyses were performed in UM supplemented with IL-1β and/or IL-6. The concentrations were determined by assaying cell viability by MTT (Supplementary Fig. 1D). As Fig. 1c,d show, these ILs, as components of the inflammatory condition, do not affect the capacity of NSCs to proliferate as no changes were detected in the neurophere’s diameter under the assayed conditions.
Effect of inflammatory stress on neuronal differentiation of NSCs. Neuronal differentiation is key in neural tissue regeneration after injuries19. To investigate this process under pro-inflammatory conditions, we analysed neuronal differentiation by immunocytochemistry using βIII-tubulin and MAP2 as neuronal lineage markers (Supplementary Fig. 2C). After culturing NSCs as neurospheres (two passages of 7 days each), cells were incubated for 3 days in media supplemented with macrophages-activated media (AM-20% V/V) as a condition of moderate inflammatory damage (Supplementary Fig. 1C), media without LPS activation (UM-20% V/V) or control media. The quantification analysis demonstrated that in the presence of AM there is a significant increase in the percentage of βIII-tubulin positive cells in comparison with the UM or the control (Fig. 2a,b). However, no changes were observed in astrocyte differentiation (data not shown). Similar results were observed at shorter time points (Supplementary Fig. 2A,B). However, a morphological observation revealed that neurons incubated with AM display a different shape than neurons at the control or neurons incubated with UM, with appar-ent tubulin disorganization, dystrophy with increased soma size, presence of vacuoles and poor development of dendritic spines (Fig. 2c,d, 4)20,21. These cells expressed neuronal markers βIII-tubulin and MAP2 and are negative for the astrocyte marker GPAF (Fig. 2c,d and Supplementary Fig. 2D). We therefore hypothesized that neuronal differentiation could be aberrant under pro-inflammatory stress, leading to cell dystrophy. To deter-mine whether the observed effect is a direct action of LPS or the inflammatory components (IL-1β and IL-6), we evaluated neuronal differentiation in the presence or absence of each molecule individually. As Fig. 3 shows, treatment with UM supplemented with the indicated concentration of LPS, IL-1β and/or IL-6 did not affect the rate of neuronal differentiation nor the morphology of the neurons.
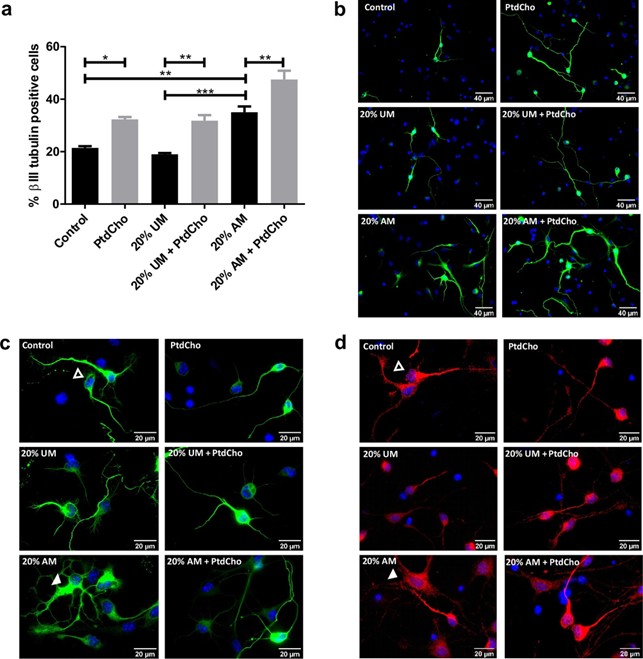
Figure 2. NSCs differentiation is affected by inflammation and restored by PtdCho. (a) Percentage of β-III tubulin positive cells analysed by immunocytochemistry coupled to fluorescence microscopy of NSCs exposed to 20% V/V of AM and UM in the presence or in the absence of PtdCho (50 μM) during 3 days. Graph represents the percentage of neuronal differentiation measured in five independent experiments. Data were presented as mean ± SEM. ***p < 0.001; **p < 0.01; *p < 0.05. (b) Representative images (× 40) of the immunofluorescence assays with the neuronal marker (β-III tubulin, green) nuclei (DAPI, blue). Scale bars: 40 μm. Representative images of neurons morphology under the indicated condition (× 100) βIII-tubulin (green) (c) or MAP2 (red) (d) Open arrow indicates normal neuron and white arrow indicates dystrophic neuron. Scale bars: 20 μm.
Phosphatidylcholine enhances neuronal differentiation and ameliorates neuronal alterations caused by inflammatory conditions. We have previously demonstrated that PtdCho, as liposomes sup-plemented in the media, regulates the fate of post-mitotic precursor cells, inducing neurogenesis15. To test the effect of this molecule under pro-inflammatory stress conditions, we incubated NSCs under each condition in the presence of egg-source PtdCho (50 µM) and counted the resulting βIII-tubulin expressing cells. As shown in Fig. 2a,b, the pro-neurogenic effect of PtdCho shown in normal and UM control conditions, is also observed under inflammation, reaching the highest levels of neuronal differentiation. Interestingly, the aberrant pheno-type observed in AM was ameliorated when cells were incubated in the presence of PtdCho (Fig. 2c,d, right panel). A detailed morphometric analysis performed after 3 and 7 days, demonstrated that the soma size, the level of βIII-tubulin expression and the presence of dendritic spines that have long been consider to provide the morphological bases for synaptic plasticity are recovered after PtdCho treatment (Fig. 4). More interestingly, the percentage of dystrophic neurons, decreases with PtdCho treatment, resulting in a significant increase in the phenotypically normal neurons (Fig. 4b) and suggesting that under inflammatory conditions, PtdCho not only regulates the fate of post-mitotic cells increasing neuronal differentiation15, but also rescues the dystrophic neurons, turning its morphology back to the normal.

