The Role of Lipopolysaccharide-Induced Cell Signaling in Chronic Inflammation
Martin J. Page1 , Douglas B. Kell1,2,3, and Etheresia Pretorius1 January 2022
Abstract
Lipopolysaccharide (LPS) is the main structural component of the outer membrane of most Gram-negative bacteria and has diverse immunostimulatory and procoagulant effects. Even though LPS is well described for its role in the pathology of sepsis, considerable evidence demonstrates that LPS-induced signaling and immune dysregulation are also relevant in the pathophysiology of many diseases, characteristically where endotoxaemia is less severe. These diseases are typically chronic and progressive in nature and span broad classifications, including neurodegenerative, metabolic, and cardiovascular diseases. This Review reappraises the mechanisms of LPS-induced signaling and emphasises the crucial contribution of LPS to the pathology of multiple chronic diseases, beyond conventional sepsis. This perspective asserts that new ways of approaching chronic diseases by targeting LPS-driven pathways may be of therapeutic benefit in a wide range of chronic inflammatory conditions.
Box 1 | Origins of LPS in the body (located at the end of the article in original form)
LPS can gain entry into the body by various routes. The majority of LPS exposure arises from translocation across the gut barrier (see166). The gastrointestinal tract has many features that restrain the microbiota while maintaining a symbiotic relationship (see167). Disruption to the intestinal barrier and increased permeability of the gut lining enables pathogens (such as bacteria), antigens and toxins to enter the bloodstream, a state referred to as ‘leaky gut’. This can be caused by inflammatory changes that occur in various diseases (see168), and is also closely associated with gut dysbiosis, an imbalance of gut microbiota (eg169). Dysbiosis is implicated in the degradation and control of tight junction proteins that govern permeability of intestinal epithelial cells (eg170), as well as immune dysregulation and inflammation in the intestine (see171,172). Further to this, diet, environmental stress, drug overuse, and conditions such as malnutrition or constipation may also lead to disruptions in gut barrier function and increased intestinal permeability.166,167
The bacterial product LPS has specifically been shown to translocate across the intestinal barrier and contribute to disease. LPS induces an increase in tight junction permeability through TLR4-dependent mechanisms,173–175 and contributes to immune activation and inflammation that further disrupt the gut barrier. Lipid absorption by chylomicrons also function as a vehicle for LPS entry.13 Direct uptake of LPS may also be mediated by M cells overlaying Peyer’s patches and by dendritic cells (see3).
LPS may further enter the circulation at other locations, such as across compromised barriers at sites of infection. For example, LPS-induced lung inflammation is linked to increased epithelial permeability in the respiratory system.176 Urinary tract infections can also be a source of bacterial molecules in the blood,177 as may medical equipment such as catheters and prosthetic devices.178 Another major source of translocated bacteria and their products is the oral cavity (see179), with epithelial barriers being disrupted with abrasive toothbrushing, or periodontal disease and associated inflammation.180 A further source of LPS is cigarette smoking (eg181).
Introduction
Considerable research into the impact of the microbiome on human health strongly implicates a role for microorganisms, their molecular products, and the host defences against them in the complex aetiopathogenesis of a broad range of chronic inflammatory diseases.1,2 Various bacterial compo-nents are recognised by the innate immune system. One of the most well studied of these is lipopolysaccharide (LPS; also known as endotoxin) that derives from the cell wall of Gram-negative bacteria such as Escherichia coli, Salmonella species, Porphyromonas species and Helicobacter species. Once in the circulation (known as endotoxaemia) LPS has a marked stimulatory effect on the immune system, and LPS-mediated signaling can cause a range of pathology.3 In severe cases this can lead to sepsis.4
Although much of the research into the physiological effects of LPS has been carried out in the setting of sepsis, LPS signal-ling is highly relevant to the pathophysiology of many chronic inflammatory diseases, including neurodegenerative disease, metabolic disease and cardiovascular disease, and particularly at LPS levels that sustain low-grade non-resolving inflamma-tion.3 LPS should therefore be regarded in chronic diseases, even if it is present in the body at only low concentrations.
In light of these newly appreciated disease associa-tions, we reappraise the roles of LPS signaling in the pathology of chronic diseases. We consider the structural aspects of LPS that are relevant to the immune response and how LPS, together with its effectors, influence immune signaling. We discuss the consequence of LPS-induced signaling in chronic inflammatory diseases and summarise the therapeutic knowledge generated from sepsis research that might be repositioned to manip-ulate LPS-driven pathways in a broad range of chronic, inflammatory diseases.
LPS: a Bacterial Wall Component
LPS is the main component of the outer membrane of most Gram-negative bacteria. LPS is composed of a lipid A hydro-phobic anchor, a proximal non-repeating oligosaccharide core, and a distal chain that is typically attached to a polymer of repeating saccharide subunits called the
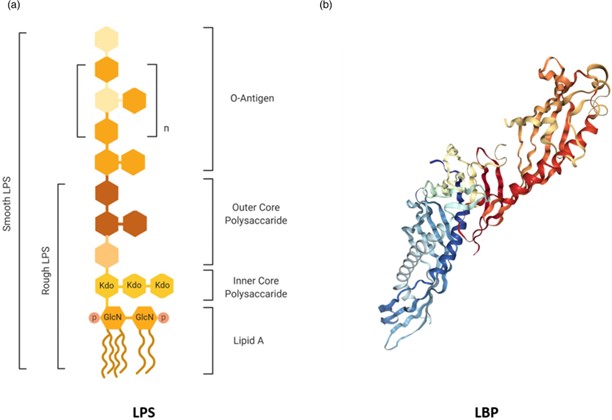
Figure 1. The structures of LPS and LBP. (a) Simplified structure of LPS from Gram-negative bacteria. The immunostimulatory lipid A is bound to a conserved inner core of sugars such as Kdo, which is bound in turn to a more variable outer core of common hexose sugars (eg glucose and galactose). The highly diverse O-antigen at the distal region of the LPS molecule is composed of repeated units of common hexose sugars. Rough-type LPS lacks an O-antigen. (b) Crystal structure of LBP (Protein Data Bank: 4M4D). The N-terminal portion is the main binding sites for lipid A, with the C-terminal involved in LPS transfer.
O-antigen that extends further from the bacterial surface (eg5,6) (Figure 1a). LPS is a potent stimulator of innate immu-nity in the host, with the lipid A moiety having most of the immunostimulatory activity (eg7,8). Structural variation of lipid A can alter the stimulatory capacity of LPS, which is related to patterns of acylation and phosphorylation and the presence of charged groups.8 Diversity in the polysaccharide chain also modulates the potency of LPS, and defines the ser-otype of the bacterium.6
Once LPS is synthesised and exported to the bacterial outer membrane, small amounts of LPS (membrane frag-ments) may be liberated from the bacterial surface during replication; larger amounts are set free during death or lysis. LPS is also secreted from bacteria through the pro-duction of outer membrane vesicles (OMVs).9 As LPS is an amphiphilic molecule, it can form micelles (supramole-cular aggregates) above a critical concentration, which varies depending on the polysaccharide chain length.10
Typically, LPS is not considered intrinsically toxic as the endotoxic effects are primarily mediated through acti-vation of the immune system (eg11). The sensitivity to LPS depends primarily on factors altering the susceptibility of the host rather than the actual mechanisms of LPS.
LPS Sensing and Cell Activation
TLR4 Activation and the LPS Transfer Cascade
LPS that gains access to the bloodstream, for example from the gut lumen (BOX 1), is bound by various particles. The major-ity of LPS (60%) circulates bound to high density lipoprotein (HDL), but also with lower affinity to other lipoproteins.12 LPS can additionally be incorporated into chylomicron parti-cles.13,14 Circulating LPS is sensed extracellularly by Toll-like receptor 4 (TLR4) in complex with MD2 (also known as LY96), which is expressed on several cell types, par-ticularly immune cells. LPS is transferred to the TLR4–MD2 complex at the cell surface by the accessory proteins LBP and CD14, which is a crucial step in mediating LPS recogni-tion by the immune system15 (Figure 2). This LPS transfer cascade massively amplifies the host response by disaggregat-ing LPS micelles (which are relatively inert and induce onlymoderate immune responses) and by catalysing the formation of complexes that activate TLR4 signaling.
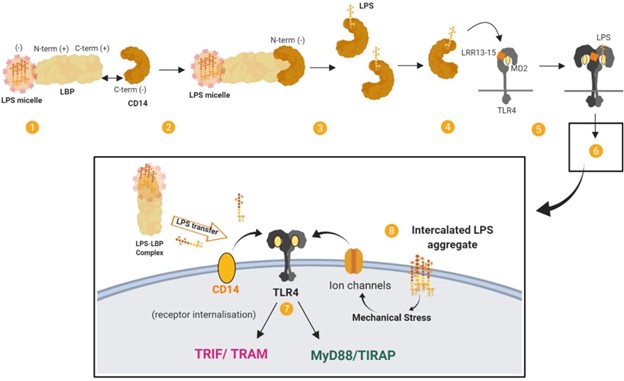
Figure 2. The LPS transfer cascade. The transfer of LPS to TLR4–MD2 through LBP and CD14 massively amplifies the host response to extracellular LPS. (1) LBP binds longitudinally to a LPS micelle. (2) Electrostatic interactions form between LBP (bound to LPS) and CD14. (3) LPS is transferred to CD14 in multiple rounds of transfer. (4) LPS is transferred from CD14 to MD2 by interacting with the LRR13–LRR15 domain of TLR4. (5) The receptor complex of TLR4–MD2 and LPS dimerises. (6) This results in signal activation by TLR4–MD2. (7) Signaling proceeds through MYD88-dependent and MYD88-independent pathways. (8) TLR4 cooperates with various co-receptors. LPS aggregates that have been intercalated into the plasma membrane by LBP may induce mechanical stress on transmembrane proteins and ion channels and contribute to LPS signaling. Adapted from.165
(3) LPS is transferred to CD14 in multiple rounds of transfer. (4) LPS is transferred from CD14 to MD2 by interacting with the LRR13–LRR15 domain of TLR4. (5) The receptor complex of TLR4–MD2 and LPS dimerises. (6) This results in signal activation by TLR4–MD2. (7) Signaling proceeds through MYD88-dependent and MYD88-independent pathways. (8) TLR4 cooperates with various co-receptors. LPS aggregates that have been intercalated into the plasma membrane by LBP may induce mechanical stress on transmembrane proteins and ion channels and contribute to LPS signaling. Adapted from.165
The TLR4–MD2 complex can be activated by structurally
diverse LPS molecules. Multiple structural components of TLR4–MD2 are involved in LPS recognition, with the highly conserved lipid A portion being an important signature for recognition by the receptor complex.16 Dimerisation of the TLR4-MD2 complex on the cell surface leads to MYD88-dependent signaling and the expression of a transcription reg-ulators such as NFκB and activator protein 1. Subsequent internalisation of TLR4 induces MYD88-independent, TRIF-dependent signaling in endosomes and the expression of reg-ulators such as interferon-regulatory factor 3, and eventually type 1 interferons. Ultimately this leads to the production of cytokines and chemokines such as IL-1β, IL-6, IL-8, CCL2 and TNF-α from MYD88-dependent pathways and CXCL10, CCL5, IFN-β and nitric oxide from MYD88-independent, TRIF-dependent pathways (see17).
The Stimulatory Functions of LBP
LBP is an important accessory protein in the LPS transfer cascade (eg18,19 and see.15,20,21). LBP is an acute phase protein produced by hepatocytes as well as in non-hepatic tissues such as the epithelial cells of the intestine, lungs, and gingival tissue. LBP is constitutively secreted by hepatocytes into the blood-stream and circulates at low levels, but can increase from a base-line concentration of around 1 to 15 µg/mL by more than 10- to 20-fold during the acute phase response to infections.22,23
LBP can form high-affinity complexes with LPS, with a specificity for the lipid A moiety24 (Figure 1b). LBP can extract LPS from LPS micelles (eg25,26) and bacterial mem-branes,27 or LPS-rich particles like OMVs, and transfer the LPS monomers to various targets such as lipoproteins, acces-sory molecules and lipid domains. LBP also has an intrinsic capacity to bind to macrophage membranes in a dose-dependent manner, independently of LPS, CD14 and TLR4.10,28 Membrane-bound LBP can act as a fusion protein and enable LPS to intercalate into liposomal mem-brane (see10). It has been proposed that membrane-bound LBP might have a role in cell activation. For example, LBP-mediated intercalation of LPS may induce mechanical stress on transmembrane proteins and stimulate K+ channels that are related to macrophage activation, with intercalated LPS aggregates being more activating than monomeric LPS.
Coreceptors and Chaperones
TLR4 can cooperate with various co-receptors, and many other molecules are involved in LPS-induced TLR4 signaling. For example, moesin is a transmembrane protein that crosslinks with the cytoskeleton and has been found to bind CD14 to form part of the complex that activates TLR4–MD2 upon LPS stimulation.29 Furthermore, ion channels such as high-conductance Ca2+ channels and voltage-dependent K+ chan-nels are activated by LPS in a dose-dependent manner and co-operate with TLR4 for cell activation. Here, voltage-dependent K+ channels are especially important for the NF-κB-dependent inflammatory response to LPS.30
In terms of chaperones, extracellular high mobility group box 1 (HMGB1), like LBP, functions as an LPS transfer molecule and catalyses the transfer of LPS mono-mers from LPS aggregates to CD14 to initiate TLR4-mediated signaling.31 HMGB1 binds to the poly-saccharide and lipid A moieties of LPS via domains located in the A and B box domains of HMGB1, respec-tively.32 HMGB1 works in concert with LPS to trigger TRIF-dependent immune responses via TLR4; down-stream regulators such as receptor-interacting protein kinase 3 (RIPK3) mediate processes such as non-resolving inflammation.33 Furthermore, HMGB1 is a ligand of receptor for advanced glycation end products (RAGE). HMGB1 acts with LPS to promote the phosphorylation of p38 MAPK and the activation of NF-κB through RAGE and thereby enhances the proinflammatory activity of LPS.34 LPS actually stimulates the release of HMGB1 from cells such as hepatocytes in a process involving TLR4, caspase-11, and gasdermin D (GSDMD).35,36
As the function of HMGB1 suggests, cell activation can occur independently of the standard LPS transfer cascade involving LBP. For example, CD14 is dispensable for cell activation at high concentrations (100-1000 ng/mL).37 Rough serotype LPS in particular can be recognised by the TLR4/MD-2 complex without the help of either LBP or CD14,38 and this direct receptor activation by rough LPS leads to efficient signaling via the MYD88-dependent pathway.39
Intracellular Sensing
In addition to its expression on the surface of various cells, TLR4 can reside on components of the endosomal systems (eg recycling endosomes, the Golgi apparatus or endoplas-mic reticulum) and detect intracellular LPS independently of cell surface TLR4.40 However, LPS can also trigger TLR4-independent responses upon intracellular detection (Figure 3).
LPS can access the cytoplasm by being transported into the cell via membrane-bound LBP or via HMGB1 and RAGE or by originating from the endocytosis of extracellular bacteria or their OMVs. LPS that accesses the cytoplasm is recognised and activates the non-canonical inflammasome pathway (see41).
In one delivery pathway, LBP interacts with the cell mem-brane independently of CD14 and TLR4-MD2 and is subse-quently internalised, which offers a route for intracellular LPS trafficking.28 In addition to transferring LPS into the cytosol, LBP is found located adjacent to activated caspases. This presents a potential mechanism for the delivery of LPS to intracellular pattern recognition receptors and the nonca-nonical inflammasome.
As a second means of entry, extracellular HMGB1 medi-ates the translocation of LPS into the cytosol and facilitates caspase-11-dependent pyroptosis.35 Specifically, HMGB1 enhances LPS internalisation into endo-lysosomes via RAGE. HMGB1 then destabilises the lysosomal membrane, allowing LPS to leak into the cytosol and activate caspase-11.
In a third pathway, pathogens and OMV can be brought into the cell through endocytosis. Pathogen-containing vacuoles (or phagosomes) carry bacterial cargo into the cell and are disrupted by interferon-inducible guanylate-binding proteins (GBPs) and IRGB10.42,43 Lysis of these vacuoles releases bacteria into the cytosol. However, GBPs do not play a role in the accessibility of OMVs to the cytosol.44 Instead, OMVs release LPS into the cytosol from early endosomes.45 The exact mechanism of this release is unknown, although haemolysin can induce the rupture of OMV-containing vacuoles.46 GBPs also facilitate the sensing of cytoplasmic LPS (and OMVs44,47) by functioning as cofactors for the activation of the noncanonical inflammasome.48 For Gram-negative bacteria that enter the cytosol, GBPs assemble on their surface and create a platform for caspase-4 activation.49,50 Caspase-4 and/or caspase-5 (caspase-11 in mice) is the primary component of the noncanonical inflammasome and acts as a cytosolic receptor for LPS.51–53 This caspase-11 pathway intersects with the extracellular TLR4 pathway. For example, the TRIF pathway, downstream of TLR4, mediates the release of type I interferons that induce caspase-11 expression54 as well as the expression of GBPs, and is crucial for OMVs-induced caspase-11 activation.55 Caspase-11 also synergises with the assembled canonical NLRP3 inflammasome pathway to regulate caspase-1 pro-cessing and IL-1β and IL-18 secretion.56
An outcome of caspase-11 activation is the initiation of pyroptosis. Pyroptosis plays a role in the clearance of intra-cellular bacteria from macrophages, which exposes patho-gens to neutrophils. GSDMD is an essential effector of pyroptosis (see57,58). GSDMD is cleaved by caspase-11 and forms pores in the cell membrane that lack ion sensitivity and compromise the integrity of cellular membranes.59–62 Cleaved GSDMD also triggers NLRP3-dependent activation of caspase-1.59 Another upshot of this process is that NLRP3-dependent IL-1β and IL-18 could possibly exit the pyroptotic cell through the GSDMD pores.61,62
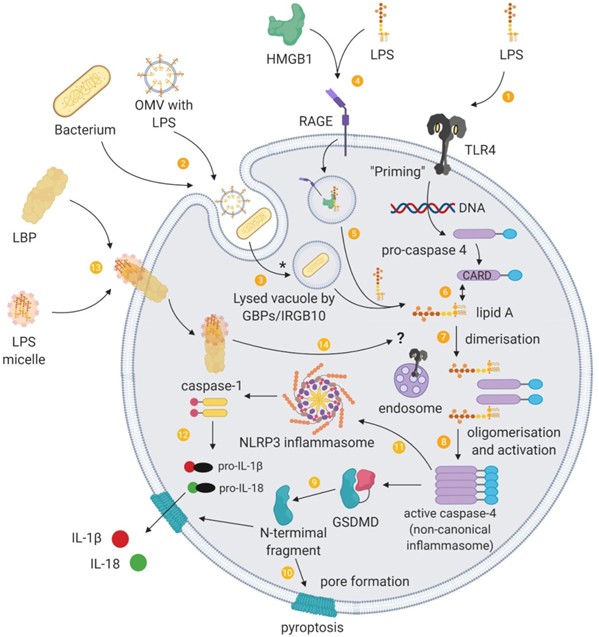
Figure 3. Intracellular sensing of LPS and the non-canonical inflammasome. (1) Binding of PAMPs such as LPS to extracellular pattern-recognition receptors activates signal transduction cascades that induce gene expression of the components required for non-canonical inflammasome activation, a process termed “priming”. (2) Extracellular Gram-negative bacteria or their outer membrane vesicles (OMVs) can deliver LPS into the cytoplasm of host cells through endocytosis. (3) Guanylate-binding proteins (GBP) together with IRGB10 disrupt vacuoles containing pathogens, which enables LPS to be released into the cytosol. (*) LPS from OMVs are released from early endosomes, which does not involve GBPs/IRGB10 mediated lysis. (4) HMGB1 promotes the internalisation of LPS into vacuoles through RAGE. (5) HMGB1 additionally permeabilises this vacuole to release LPS into the cytosol. Cytosolic LPS can be recognised by inflammatory caspases and to provide a “triggering” signal for activation of the non-canonical inflammasome pathway. (6) The lipid A tail of LPS binds to the CARD motif of pro-caspase 4 in a direct interaction. (7) These intracellular LPS–pro-caspase 4 complexes dimerise and (8) oligomerise through the CARD motif, and proximity-induced activation leads to a catalytically active conformation of the non-canonical inflammasome. (9) The activated non-canonical inflammasome cleaves gasdermin D (GSDMD), releasing the amino-terminal fragments, (10) which form pores at the cell membrane. (11) The non-canonical inflammasome also activates the NLRP3 canonical inflammasome through an unknown mechanism, (12) and this inflammasome pathway releases IL-1β and IL-18, which is secreted through GSDMD pores. (13) LPS can additionally be delivered into cells through cell-bound LBP. (14) Intracellular transport of LPS by LBP raises the possibility of a role for LBP in the delivery of LPS to intracellular LPS receptors such as endosomal TLR4 and inflammatory caspases.
Functions of LBP in Response to LPS
LBP does not only facilitate the transfer of LPS to the TLR4 complex to maximise LPS-induced signaling (see20,21) but has multiple functions in lipid transfer and immune regulation for the recognition and control of bac-terial infections. The role of LBP in these responses is con-centration dependent: at low concentrations LBP potentiates LPS-induced immune activation, whereas with the acute-phase rise in LBP concentration LBP inhib-its LPS-induced cellular stimulation and promotes LPS clearance to protect the host from bacterial challenge (eg63,64). As the potency of LPS is due to the sensitivity of the host recognition system, LBP has a crucial role in modulating the effects of LPS. Thus, LBP not only induces LPS responses but also terminates them (although these functions are more complex in diseases).
The Inhibitory Functions of LBP]
LBP, especially at acute phase concentrations, exhibits various inhibitory actions that attenuate the stimulatory effects of LPS23 (Figure 4). LBP can dissociate LPS from membrane-bound CD14 and inhibits the transfer of LPS from soluble CD14 and soluble MD2, thus attenuating cell signaling.64,65 LBP also promote the cellular uptake of LPS, which reduces the capacity of LPS-induced signaling through TLR4.64 However, LPS transfer or cellular uptake facilitated by LBP is likely not the most important mecha-nism for clearing LPS from the blood.66
The inhibitory action of LBP also depends on the ability of LBP to transfer LPS to other targets including lipoproteins such as HDL27,67; a process that neutralises LPS, particularly by interfering with the internalisation of LPS by macro-phages.68 Transfer of LPS to HDL also facilitates the removal of LPS in the liver69, and LBP further mediate LPS detoxification in the liver by enhancing binding to chy-lomicrons in a dose-dependent manner.14
Additionally, LBP intercalated into LPS aggregates can mediate the crosslinking of several layers of lipid A, which has a neutralising effect by decreasing the accessibility of lipid A.70 This complexation increases the binding energy of the LPS in the complex as well as prevents LBP from inserting in the plasma membrane. LPS–LBP complexes in the plasma do not interact with membrane-bound LBP, which limits some of the stimulatory mechanisms of LPS.10
Furthermore, phospholipids can inhibit the binding of LPS aggregates to LBP, probably through competitive binding, and also reduce LBP-mediated transport of LPS to the cell membrane, attenuating cell activation.71 LBP addi-tionally facilitates the binding of phosphatidylinositides and phosphatidylserine to membrane-bound CD14 and this inhib-its LPS-induced responses in monocytes by competing for binding with LPS on CD14.72
The Double-Edged Sword of LBP
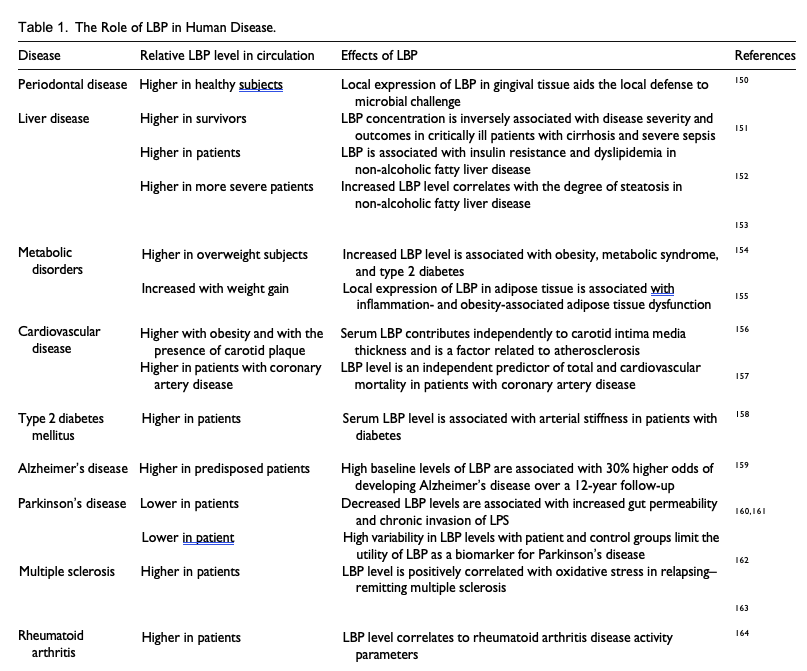
The effects of LBP levels in disease are highlighted in Table 1. LBP is usually seen as a host-protective factor to manage the effects of LPS.73 It enables the immune system to respond efficiently to bacterial invasion but also exhibits a dual function by blunting these effects at high doses. However, the activity of LBP in enhancing LPS-mediated stimulation and cytokine release is also viewed as a contributor to the pathogenic state mediated by LPS. In mice, antibody-mediated depletion of LBP see-mingly protects against LPS-induced toxicity,74 possibly even attenuating the onset of disease (eg diet-induced non-alcoholic fatty liver disease in mice75) and dysfunction (eg endothelial dysfunction and tissue fibrosis in a swine model of LPS-induced acute kidney injury.76) Furthermore, certain LBP gene polymorphisms are associated with an increased risk of pathology. For example, a functional variant in the LBP promoter (SNP 1683) has greater promoter inducibility, which is associated with higher median basal serum levels of LBP and a 5-fold increase in Gram-negative bacteraemia-related mortality of patients after allogeneic hematopoietic cell transplantation.77 Acute-phase levels of LBP can also potentiate rather than inhibit overwhelming inflammatory responses following LPS challenge compared with LBP knockout mice in the setting of cholestatic liver disease.78
To rehearse, the reactivity of LPS depends on the sensi-tivity of the host and the toxic effects of LPS are mediated by the host immune system. It could be said that LBP can contribute to damaging immune reactions by augmenting immune activation. Even so, a reduced immune response in the absence of LBP results in increased susceptibility to infection and its lethal effects.21,66,79 Nonetheless LBP is not simply a protective factor.
The Role of LPS in Chronic Disease
LPS-induced signaling — through TLR4-dependent and TLR4-independent pathways — evokes a profound inflammatory response and has a wide range of effects on cells (Figure 5). In addition to the release of various inflammatory mediators following leukocyte activation (see17), LPS also participates in other aspects of the immune response such as promoting dendritic cell matura-tion and migration, which links the innate immune response to adaptive immunity80,81 (the same is true for OMVs82), and promoting autophagy in macrophages through p38 MAPK dependent TLR4 signaling,83 which serves as a process that degrades sequestered pathogens. LPS also triggers the production of reactive oxygen and nitrogen species, such as through TLR4-mediated NADPH oxidase activation in macrophages,84 which leads to oxidative and nitrosative stress and damage; reactive oxygen and nitrogen species are also linked to inflammatory signaling and toxic responses.
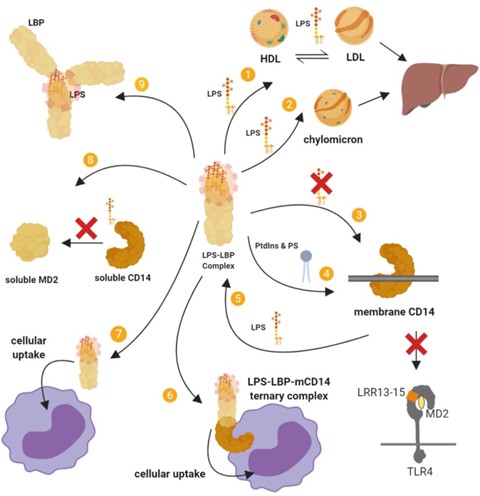
Figure 4. Summary of the inhibitory actions of high-dose LBP. (I) LBP mediates LPS transfer to lipoproteins and (2) chylomicrons, which sequesters LPS, attenuates its stimulatory effects, and leads to its intestinal excretion via the liver–bile duct pathway. (3) LBP supresses the transfer of LPS to membrane-bound CD14 (mCD14) and subsequently to TLR4–MD2. (4) LBP facilitates the binding of a series of phosphatidylinositides (PtdIns) and phosphatidylserine (PS) to mCD14, and this inhibits LPS–mCD14 binding and LPS-induced responses. (5) LBP can also cause the dissociation of LPS from mCD14. (6) The LBP–LPS complex may also remain associated with mCD14 to form a ternary LBP–LPS–mCD14 complex that does not trigger signaling and is eventually internalised. (7) Cellular uptake may also proceed by a CD14-independent pathway. (8) LBP can inhibit the transfer of LPS from soluble CD14 (sCD14) to soluble MD2 (sMD2). (9) LBP can bind and intercalate into LPS aggregates, which has an inhibitory effect. The LBP–LPS complex cannot intercalate into the membrane, cannot bind LPS into the membrane and cannot interact with membrane-bound LBP. LBP can also mediate the crosslinking of several layers of lipid A, thereby decreasing its accessibility. It has also been proposed that by opsonising LPS aggregates at high density, LBP causes steric hindrance to other molecules such as CD14.
The excessive production of inflammatory mediators such as cytokines, chemokines and acute phase proteins, including complement component C3, C-reactive protein (CRP) and LBP, can result in overwhelming stimulation of the immune response and lead to cell damage and have lethal effects.3,4,85,86 This is epitomised in sepsis where a dysregulated host response to infection culminates in a pathological syndrome that can include disseminated intravascular coagulation and multisystem organ failure.4,86 However, there are many diseases in which LPS levels are raised in patients relative to matched con-trols.3,87 Here, the effects of LPS and its downstream sig-nalling contribute to diverse pathologies.
Neurodegenerative Disease
LPS is implicated in the pathogenesis of neurodegenerative diseases such as Alzheimer’s disease, Parkinson’s disease and amyotrophic lateral sclerosis (see88). Neuroinflammation is a key pathological event in the neurodegenerative process.
Systemic inflammation is similarly an important factor and systemic exposure of animal models to LPS leads to neurode-generative pathology (see89). LPS and neurodegenerative stimuli, such as β-amyloid, tau or α-synuclein, synergise to induce neurodegeneration (see88,90). LPS may prime microglia to neurodegenerative stimuli, and conversely neurodegenera-tive stimuli may prime microglia to LPS challenge. On the other hand, LPS exposure may lead to insensitive microglia with decreased activation, which reduces protective functions in the brain such as phagocytosis of protein aggregates. Either pathway leads to stressed neurons.
Indeed, LPS treatment of mice leads to cognitive impair-ment, which is prevented by TLR4-specific inhibitors.91,92 LPS also activates microglia and disrupts synapses in a mechanism involving microglia-derived IL-1β and loss of synaptophysin in mice.93 Furthermore, LPS stimulation can increase the permeability of the blood brain barrier in aged mice.94 Although, the blood brain barrier is relatively resis-tant to LPS-induced disruption, LPS can cause disruption through both paracellular and transcytotic mechanisms.95
In the context of Alzheimer’s disease, LPS associates with amyloid plaques, neurons, and oligodendrocytes in brains of patients, and causes injury, such as to myelin.96,97 In the mouse brain, inflammation generated by systemic LPS leads to Aβ42 accumulation through increased β- and γ-secretase activities and increased expression of amyloid precursor protein, among other effects.98 Experimentally, LPS can promote fibrillogenesis of Aβ fragments and Aβ fibrils bind directly to LPS micelles.99 In mice, LPS-induced microglial activation also promotes hyperpho-sphorylation of tau through pathways involving microglial-derived IL-1 and neuronal p38 MAPK.100
Metabolic Disease
Clinical studies of metabolic endotoxaemia in obese patients have demonstrated that serum levels of LPS increase with a high-fat diet.101 High-fat meals similarly lead to increased LPS levels in non-obese102 and diabetic103 individuals. In mice, metabolic endotoxaemia dysregulates the inflamma-tory response and triggers the onset of diabetes and obesity.104 Obesity is characterized as a state of chronic low-grade inflammation and LPS-induced signaling contributes to the proinflammatory milieu in human obesity.105
LPS also promotes adipose dysfunction. LPS might be involved in defining the adipocyte death size, by initiating pyroptosis, and in the formation of crown-like structures in adipocytes, composed of dead adipocyte remnants and
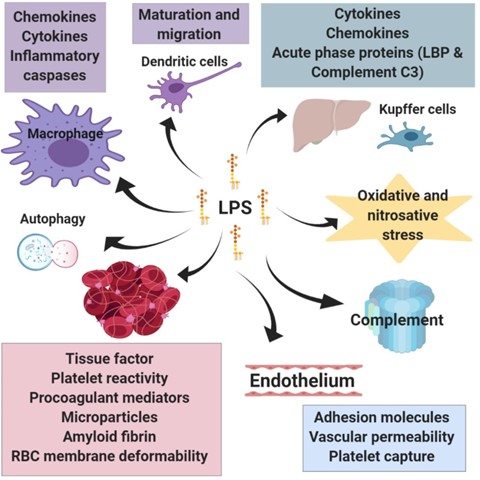
Figure 5. Summarised downstream effects of LPS signaling. LPS influences a range of cell types and physiological processes. The activation of leukocytes initiates the immune response and the release of various inflammatory mediators. LPS also activates specific immune processes such as the maturation and migration of dendritic cell, autophagy in macrophages, and activation of the complement system. In the liver, LPS stimulates the production of acute phase proteins as well as several inflammatory mediators. Similarly, LPS promotes inflammatory reactions in adipose tissue. These mediators as well as the increased activity of enzymes involved in the production of reactive oxygen and nitrogen species contribute to a cellular stress. A major action of LPS is its ability to promote coagulation both by enhancing the expression of molecules that stimulate clotting and through direct interaction with red blood cells and platelets. LPS-mediated changes to the endothelium also promote coagulation.
macrophages, as well as in promoting the transmission of macrophages to a proinflammatory phenotype (see106). Indeed, obese patients with a higher degree of metabolic endotoxaemia experience altered expression in genes for adipose tissue function and lipogenesis as well as increased expression of inflammatory markers in this tissue.107
Cardiovascular Disease
Atherosclerosis underlies the majority of cardiovascular dis-eases. LPS accelerates atherosclerosis in hypercholesterol-emic rabbits108 as well as provokes more severe atherosclerosis in mice.109 Epidemiological studies show that subclinical endotoxaemia is a strong risk factor for the development of atherosclerosis.110 Further, TLR signaling
has a prominent role in atherosclerosis (see111), with LPS being a potential source of vascular inflammation.
Experimentally, LPS triggers inflammatory responses in human vascular endothelial cells.112 In studies of human tissue and primary cell culture, LPS induces the expression of neuraminidase-1, an enzyme involved in atherosclerosis progression, in monocytes, which is also part of a positive feedback loop that stimulates the expression of proinflamma-tory and proatherogenic cytokines.113 Furthermore, subclini-cal doses of LPS in mice reduce interleukin-1 receptor associated kinase M (IRAK-M) and induce miR-24, a microRNA precursor, in monocytes, which are important for resolving inflammation and homeostatic tolerance.114 Disruption to this system sustains the non-resolving low-grade inflammatory phenotype of monocytes, which is con-ducive for the aggravation of atherosclerosis.
A further way LPS experimentally contributes to athero-sclerosis is by promoting lipid accumulation in human adventitial fibroblasts via TLR4 and by promoting the pro-duction of monocyte chemoattractant protein (MCP)-1, an important chemokine in the initiation of atherosclerotic plaques.115 LPS additionally upregulates the expression of the Fcα/µ receptor on macrophages experimentally, which promotes the binding of oxidised-LDL/IgM complexes and the formation of foam cells.116
Coagulopathy
LPS is a well-known and powerful pro-coagulant. This increases the risk for thromboembolic events, such as stoke, and can lead to more severe coagulopathy, such as dissemi-nated intravascular coagulation. LPS upregulates the expres-sion of tissue factor in a complement- and CD14-depedent manner,117 which is a crucial initiator of thrombin formation and consequently fibrin deposition. LPS can also bind to fibrinogen and induce misfolding.118,119 In addition to a role in coagulation, thrombin can cleave pro-IL-1α and initiates the IL-1α inflammatory cascade.120 Indeed, LPS-induced inflammatory processes promote coagulation (see4,86). Contrariwise coagulation factors also activate inflammatory signaling pathways, largely through protease activated recep-tors (see121). Inflammatory processes additionally downregu-late anticoagulant mechanisms such as the anti-thrombin system, tissue factor pathway inhibitor, and the activated protein C system; an increase in plasminogen activator inhib-itor type 1 also contributes to curtailed fibrinolysis.
Activation of the noncanonical inflammasome by LPS is a further pathway that induces systemic blood clotting. Pyroptotic macrophages release tissue factor in the form of microvesicles through GSDMD pores, which initiates coagu-lation cascades.122 Additionally, GSDMD-mediated calcium influx and activation of scramblase enzymes cause phospha-tidylserine exposure that increases tissue factor activity and further drives coagulation activation.123 Extracellular HMGB1 and its interaction with LPS for intracellular deliv-ery promotes this process.124 The contribution of HMGB1 to LPS-induced coagulopathy is amplified by LPS-induced type I interferon signaling downstream of TLR4 that increases the extracellular release of HMGB1.124
Furthermore, LPS, the complement system and various inflammatory mediators induce the platelet response.125 Platelet TLR4 activation is an important trigger for platelet hae-mostatic responses although the effect of LPS on platelet TLR4 is controversial (see126). OMVs additionally induce platelet-platelet aggregation and degranulation as well as platelet-leukocyte binding. Ex vivo OMVs are a more potent platelet agonist than is purified LPS.127 Platelet activation can further propagate coagulation and inflammatory signaling.128
Therapeutic Implications
Research into therapeutic strategies that target bacterial LPS has mainly been carried out in the context of sepsis. These strategies aim to either neutralise or remove LPS or inhibit LPS-mediated activation of immune cells, thus reduc-ing the dysregulated host response. However, therapies aimed at neutralising the effects of LPS by inhibiting the inflammatory response or by LPS-targeted antibodies (such as E5 and HA-1A) have been largely ineffective in treating sepsis. Nonetheless, it is worthwhile revising these therapeu-tic approaches when searching for potential therapies that target LPS in chronic diseases.
The monocolonal antibody HA-1A (marketed as Centoxin), which targets LPS, and recombinant human acti-vated protein C (Drotrecogin alfa; marketed as Xigris), an anticoagulant, are notable failures of the biopharmaceutical industry.129,130 The LPS–TLR4 signaling pathway has also been considered as a target to treat sepsis,131 but the lipid A antagonist eritoran, which blocks LPS from binding to TLR4–MD2,132 and the TLR4 antagonist TAK-242133 have been ineffective in reducing sepsis mortality. Several reasons are offered to explain the failures of these approaches including the complexity of the physiological response in stressed patients; the clinically appropriate time to administer certain drugs (eg during early exposure vs. during established pathology); heterogeneity in crucially-ill patient populations; the difficulty of standardising the timing of therapeutic inter-ventions among patients; and the presence of generic poly-morphisms within the general population that are not necessarily proportionally represented in clinical trials and potentially affect trial outcome and data analysis.134 Of course, another explanation is simply that these reactions are not part of the main pathways driving sepsis.
Targeting LPS Through Neutralising Molecules
The development of LPS-neutralising peptides is an area of active research to treat sepsis. Protonated histidine-rich polypep-tides can inhibit LPS responses by binding and neutralising LPS, although their moderate toxicity, poor solubility at physi-ological pH and immune activating effects limit immediate ther-apeutic benefit (see135). Also, their LPS-neutralising effect is tightly coupled to the protonated state, which is largely lost at physiological pH. Other polycationic peptides such as procalci-tonin136 can also neutralise LPS and reduce the expression of downstream inflammatory mediators. By contrast, arginine-and lysine-rich cationic polypeptides enhance LPS responses by shuttling monomerised LPS to sites of recognition.135
Nonetheless, peptide-based therapies based on LPS-binding domains are a promising approach to target and inhibit LPS. Peptides based on the iron-binding glycoprotein lactoferrin as well as defensins and fragments of saposin family proteins have been studied for their ability to neutralise LPS (see137,138). Synthetic anti-LPS peptides139 and Pep19-2.5140
also interact with LPS and interfere with its signaling cascade. Sushi peptides, derived from horseshoe crab, can bind LPS, compete against other binding proteins, and display detergent to like properties in disrupting LPS aggregates.141 Recombinant or synthetic host antibacterial protein fragments with high affinity for LPS, such as bactericidal/permeability-increasing protein (BPI) and 18-kDa cationic antimicrobial protein (CAP18), have been proposed as candidate agents in adjunctive therapy for Gram-negative sepsis.137,138 However, problems in terms of the timing of treatment, possible toxicity, and probable rapid clearance from the circulation might limit the use of these fragments therapeutically.
Further to peptide-based therapies, other agents also demonstrate LPS neutralising abilities. Cationic lipids prevent LPS from evoking an immune response, possibly by trapping LPS in mixed micelles that are harder to disag-gregate, thus detoxifying LPS.135 Intravenous administration of protein-free, phospholipid-rich emulsions can neutralise LPS142 but (perhaps surprisingly) these emulsions have shown no benefit in clinical trials of sepsis.143 Lipopolyamines such as oleoyamines can also neutralise LPS and its signal-ling, and are of interest due to their low toxicity, low immu-nogenicity, and ease of chemical synthesis.144 Lastly, synthetic cationic-amphiphilic polymers can detoxify LPS by binding and forming pseudoaggregates.145
Other Targets to Reduce LPS and its Effects
Additional targets for treating endotoxemia might include sites of LPS release into the circulation. The gut and other focus sites (such as periodontal disease, catheters, and vascu-lar access) are potentially reversible sources of LPS translo-cation into the circulation. Treatment may include the use of non-absorbable drugs that form complexes with LPS in the intestine and reduce translocation, such as sevelamer.146 By strengthening the intestinal epithelial barrier, nutritional management including improving dietary quality and micro-nutrient status, manipulating the gut microbiome with prebio-tics and probiotics, and using certain nutritional supplements may be alternative treatment strategies to help manage circu-lating levels of LPS and inflammation.147 Indeed, natural compounds such as Zingerone from ginger attenuate LPS-induced inflammation.148
Anti-inflammatory antioxidants may hold promise for neu-tralising the inflammatory effects of LPS, even if they do not interact with it directly. However, although using an anti-inflammatory therapy may seem a logical approach, research of sepsis has shown that extended immunosuppression (anergy) following an initial pro-inflammatory phase may be detrimental and increase susceptibility to secondary infec-tions.149 Therefore, an important consideration is to restore the balance between pro-inflammatory and anti-inflammatory reactions. This offers a further example of the complex dynam-ics of inflammation operating in diseases such as sepsis
Conclusions
LPS is increasingly found to be associated with chronic dis-eases and can cause various pathologies. Numerous mecha-nisms are proposed to link LPS-induced signaling and immune dysregulation to the aetio-pathogenesis of chronic diseases. The aetiologies and pathogenesis of these diseases are multifactorial with several factors contributing to their risk and progression. LPS signaling is presented in this Review as a potential common pathway operating across these conditions.
Chronic inflammatory diseases are collectively the most prevalent conditions affecting human health, representing some of the greatest burdens to society. According to WHO factsheets, more than 55 million people have dementia (60%-70% of which is Alzheimer’s disease) at an economic cost of $1.3 trillion in 2019. In 2014, 422 million people had diabetes, which accounted for 1.5 million direct deaths in 2019. Cardiovascular diseases are the leading cause of death globally at an estimated 17.9 million deaths in 2019, which represents 32% of global deaths and 38% of deaths classified as premature. Even small improvements to these statistics by regarding the role of LPS in chronic diseases could yield significant impact.
Traditionally, LPS research has been carried out in the context of sepsis, especially concerning the quest for thera-peutics that target LPS. However, there exists promise for the translation of sepsis research to other pathological condi-tions. As an example of translation, drugs developed for sepsis treatment that improve functional outcomes (for example, by reducing morbidities, delaying the onset of organ failure, or improving outcomes on a disease-specific performance scale) but that have not been linked to reduced 28-day mortality may still show promise in other conditions where endotoxemia is less severe. Indeed, approaches that aim to reduce levels of LPS in the blood and thus mitigate its interaction with the host may lead to reduced pathology in a broad range of conditions. However, developing treatments that can lower circulating LPS levels over the long term will be challenging. Nonetheless, a starting point is to look out for and treat gut disturbances, as gut dysbiosis can be a major source for LPS to enter the blood circulation.
Overall, research in the setting of sepsis has laid a foun-dation for understanding LPS-induced signaling and its contribution to disease processes. Future research needs to establish the clear causal links between chronic diseases and elevated LPS. Determining the effect of therapeutics that target LPS levels or its downstream actions in chronic diseases needs to form part of these future investi-gations. Some success in translation has come from the failure of the antibody drug Centoxin, where experience in developing antibody therapies has informed the subse-quent scientific planning and development of other anti-body drugs for a number of chronic conditions (see129). Appreciating the relevance of LPS-driven pathways as contributors to pathology in a range of diseases therefore offers a novel framework for approaching these conditions that might offer new solutions to the public health burden of chronic inflammatory diseases.

